Biochemistry- Wendler Research Group
Welcome to the Wendler lab @ University of Potsdam
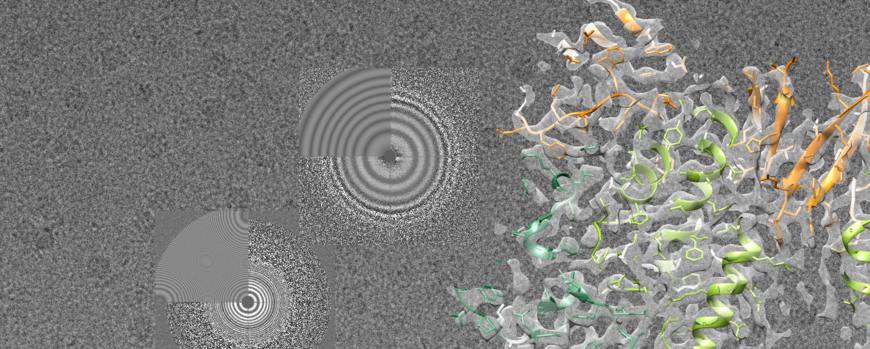
We use cryo electron microscopy and single particle analysis to understand the function of molecular machines.
Our aim is to solve the conformational dynamics of molecular machines at the highest possible resolution using single particle cryo EM. We work on different biological problems such as proteasomal biogenesis, electron transport in redox protein complexes and molecular chaperones using structural biology as well as biophysical methods. One focus in the lab is the analysis of members of the large protein superfamily of AAA+ proteins (ATPases associated with various cellular activities), which are usually at the core of many essential multi protein assemblies involved in re-organisation and recycling processes of membranes, proteins or DNA in the cell. They convert chemical energy into mechanical work, and our lab aims to obtain a structural view on the complex conformational dynamics of these fascinating molecular machines in action. In particular, we want to understand how allosteric interactions between AAA+ modules in the active oligomer regulate ATPase activity and how accessory factors influence complex activity. The ultimate goal is to integrate structural data obtained by cryo electron microscopy and X-ray crystallography with mutational analysis and biophysical experiments in order to tackle severe disorders and illnesses stemming from malfunctioning molecular machines.
Fields of interest: single particle cryo electron microscopy; catalytic enzymes that hydrolyse ATP or fixate carbon dioxide; AAA+ proteins; peroxisomal protein import; electron transfer in biological systems, redox proteins; proteasomal biogenesis.