Endocardial chamber morphogenesis
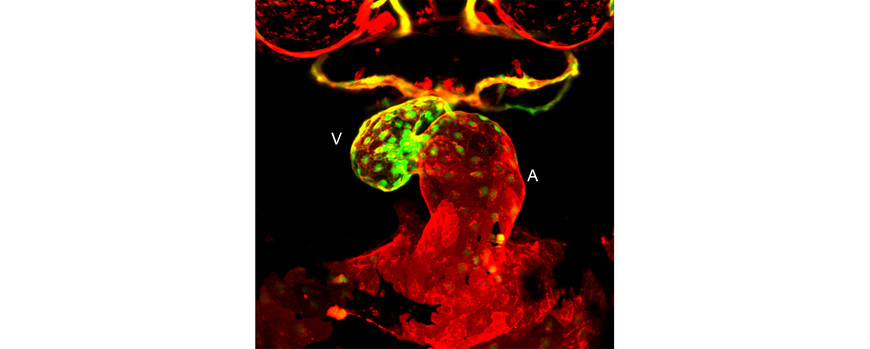
In another study, we analyzed the process of chamber morphogenesis of the endocardium, a specialized endothelial tissue that lines the interior of the heart (Dietrich et al., Developmental Cell 2014). During heart development, the onset of heartbeat and blood flow coincides with a ballooning of the cardiac chambers (Figure 2). By combining functional manipulations, fate mapping studies, and high-resolution imaging, we showed that endocardial growth occurs without an influx of external cells. Instead, endocardial cell proliferation is regulated, both by blood flow and by Bmp signaling in a manner independent of Vegf signaling. We also found that during cardiac ballooning stages, endocardial cells obtain distinct chamber- and inner-versus-outer-curvature-specific surface area sizes. Finally, we showed that the hemodynamic-sensitive transcription factor Krüppel-like factor 2a (Klf2a) is an important regulator of endocardial cell morphology. These findings establish the endocardium as a bood flow-sensitive tissue in the heart with a key role in adapting chamber growth in response to this mechanical stimulus.
Taken together, our findings are indicative of dynamic interactions between the myocardium and endocardium that constantly attune cellular sizes and shapes, and thus cardiac chamber dimensions in response to physiological changes, the most important of which is blood flow. Understanding this dynamic crosstalk during development may have implications for understanding how cardiac morphology changes later in life, in response to physiological adaptations or as pathophysiological conditions arise.
Figure 2: Frontal view shows the two distinct endocardial chambers, atrium (A) and ventricle (V), during cardiac ballooning stages in a 48 hour old zebrafish embryo. The two endocardial chambers are visualized with the transgenic reporter lines [Tg(flt1:YFP), green; Tg(kdrl:mcherry), red].