Welcome
Cell2Fab - from cells to fabrication
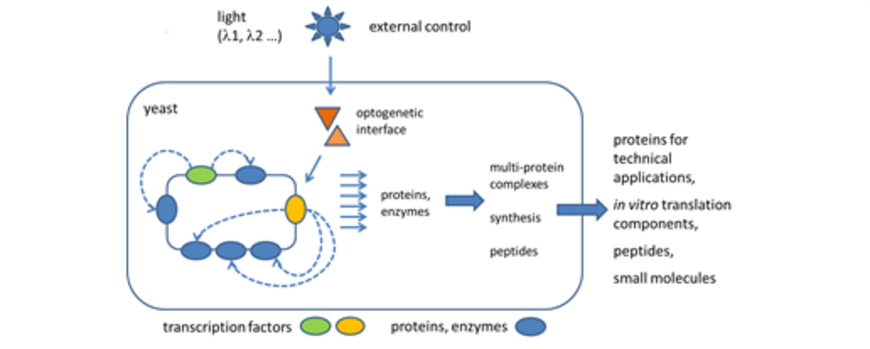
The project aims at the generation of a novel cellular regulation system on the basis of circular yeast chromosomes (xYAC) thereby facilitating highly regulated production of small to large cohorts of proteins and peptides in the yeast Saccharomyces cerevisiae. Initially, the basic structure of the xYACs will be designed to allow straightforward and effortless integration of gene regulatory elements and the versatile application of xYACs for next generation biotechnology.
Several different regulatory elements with distinct regulatory characteristics will be designed including the later integration of optogenetic regulation systems into the xYACs. The long-term goal is the use of xYACs for the generation of “multi-enzyme machines” for the production of pharmacologically relevant peptides, entirely new functional units for next generation biotechnological procedures or the production of proteins for in vitro translation. xYACs will be designed as open system allowing further development to ensure fast and adequate adaptation to the needs of future users.

Kontakt
Dr. Katrin Messerschmidt
Nachwuchsgruppenleiterin