Intrinsically Disordered Proteins - Research
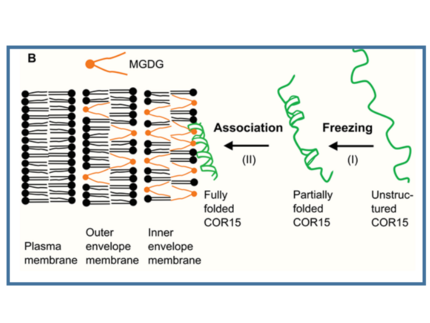
Our model proteins are LEA (late embryogenesis abundant) proteins, a group of IDPs related to cellular dehydration. They accumulate in seeds and vegetative plant tissues, especially after exposure to abiotic stresses and in desiccation tolerant bacteria and invertebrates. Most LEA proteins are intrinsically disordered under fully hydrated conditions and fold during drying. While literature agrees on a protective function of LEA proteins during desiccation and dehydration on a physiological level, their molecular functions are still largely unknown. In vitro, several LEA proteins are capable to stabilize membranes or enzymes during mild dehydration or full desiccation.
Our general interest lies in understanding the molecular functions of LEA proteins and how these are related to conformational flexibility. Further, we aim to elucidate how functional specificity of LEA proteins is encoded in their primary sequence.
We are using a combination of biophysical, biochemical and computational approaches. Our central methods are cloning, protein expression and purification, spectroscopic (circular dichroism, infrared and fluorescence spectroscopy) and hydrodynamic (static and dynamic light scattering) methods and classical protein biochemistry.