Functions and regulation of V-ATPase
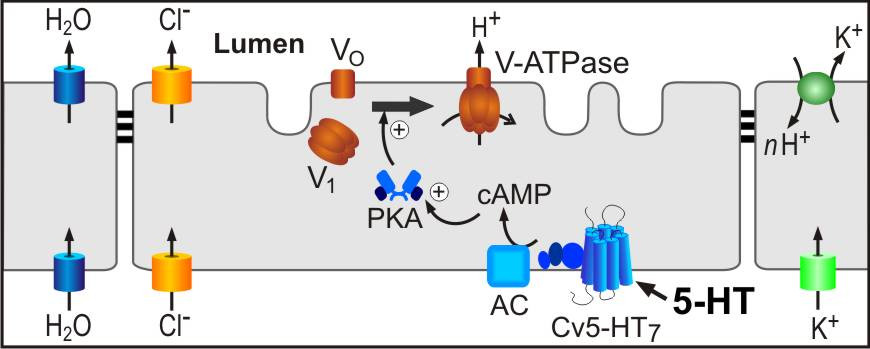
The vacuolar-type proton pump (V-ATPase) uses the energy of ATP hydrolysis for the electrogenic transport of protons across membranes. V-ATPase is organized in two sectors, a membrane-integral VO domain that conducts protons across the membrane, and a cytosolic V1 domain that contains the ATPase activity. Various mechanisms have been implicated with the regulation of V-ATPase activity, e.g. reversible disassembly of functional V1VO holoenzymes into inactive VO and V1 domains.
We use the salivary gland of the blowfly (Calliphora vicina) as a model system to determine how assembly/disassembly of V-ATPase is regulated. In this organ, the neurohormone serotonin (5-HT) elicits production of a KCl-rich saliva, and this process is energized by V-ATPase-dependent H+-transport and hyperpolarization of the apical membrane. 5-HT-induced activation of V-ATPase goes along with an assembly of V-ATPase holoenzymes on the apical membrane of the secretory cells. Using optophysiological, electrophysiological, fluorescence microscopic and molecular biological techniques, we have disclosed the signaling sequence between 5-HT receptors on the basolateral membrane and V-ATPase assembly on the apical membrane (see scheme below).
selected publications:
- Baumann O, Walz B (2012) The blowfly salivary gland - a model system for analyzing the regulation of plasma membrane V-ATPase. J. Insect Physiol. 58, 450-458
- Rein J, Voss M, Blenau W, Walz B, Baumann O (2008) Hormone-induced assembly and activation of V-ATPase in blowfly salivary glands is mediated by protein kinase A. Am. J. Physiol. Cell Physiol. 294, C56-C65
- Voss M, Vitavska O, Walz B, Wieczorek H, Baumann O (2007) Stimulus-induced phosphorylation of V-ATPase by protein kinase A. J. Biol. Chem. 282, 33735-33742
- Dames P, Zimmermann B, Schmidt R, Rein J, Voss M, Schewe B, Walz B, Baumann O (2006) Cyclic AMP regulates plasma membrane V-ATPase assembly and activity in blowfly salivary glands. Proc. Natl. Acad. Sci. USA 103, 3926-3931